
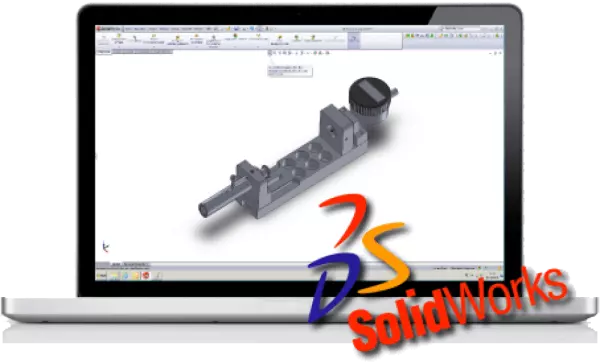
CAD construction / development of medical...
CAD construction / development of medical products CAD has supplanted the drawing board and many...
Portal and digital medical technology fair of the largest MedTech cluster in Germany

CAD construction / development of medical...
CAD construction / development of medical products CAD has supplanted the drawing board and many...

Research & development in the field of...
Do you have a product idea in the field of medical technology and need support with development?...

All of our new developments are carried out with Solidworks. Extensive calculations, such as strengt...

Our experience ranges from all kinds of written material for clinical studies (recruitment, marketin...

Medical technology industrial design
Medical technology Industrial design is an integral part of our product design process. We strive to...

Orthopedic Products - Inserting Tools
The product range of cutting tools in the field of orthopedics includes: Phillips blades...

The classic instrument construction, paired with modern CNC technology is the goal and endeavor of m...
Contract manufacturing of precise turned...
Contract manufacturing of precise turned and milled parts for medical technology perame...

Medical technology engineering & development
Innovative and creative product design is vital to any successful product. For us it is important to...

KeyShot is the easiest-to-use 3D rendering and animation software out there. In just a few steps you...

Experience in fixture construction
In order to be able to position series or prototypes on processing machines safely and with repeatab...

IVD Engineering Solutions - Custom Solutions
DIALUNOX Engineering Services - You have the Idea - we develop your product Custom Solutions D...

Contract Packaging and Sterilization for...
Contract Packaging and Sterilization of Medical Products From raw material to ready-to-...

Healthcare & Medical assemblies
We are the reliable partner covering all areas from conception to (sterile) packaging of the finishe...

From the beginning in every JakuTec bespoke project, we work in close consultation and define prec...

Patent translation Your intellectual property is valuable. We help you to protect it internationa...

In order to be able to transport, store, clean and sterilize high-quality implants and instruments i...

Up to date technology allows us to manufacture the most complex items even in low quantities. &nb...

Production and design of packaging for medical...
PACKAGING DESIGN Blister design Along with standard blisters for product packaging and transpo...

Medical technology translations
Medical technology translations Our communication specialists have linguistic as well as technica...

Healthcare & Medical Advanced Consumables
We produce smart medical consumables with added value. Added value means optimized and expanded prod...
Case Study: Arterial Blood Collection Vessels
Whole blood samples are taken from patients using arterial blood collection vessels. The sample is t...
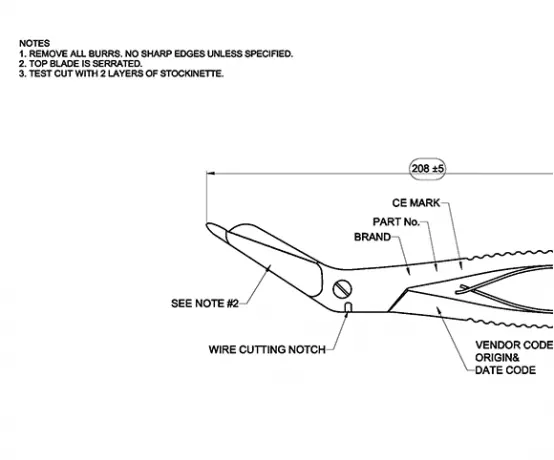
All quality-relevant data is shown centrally on the respective drawing. It can be a 2D or 3D drawing...
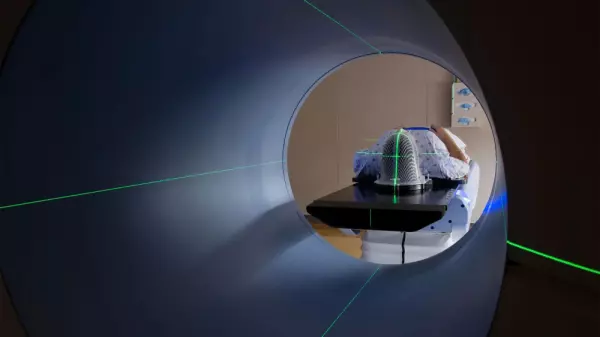
Development of Medical Devices - Systems...
Software Development We work in each phase of the medical devices’ development lifecycle an...

Orthopedic products - Navigating tools
Customized tools for navigating the drill holes and saw cuts

Time equals money when turning an idea into a marketable medical device. To accelerate your journey...

DEVELOPMENT OF SURGICAL INSTRUMENTS
CUSTOMIZED INSTRUMENTS DMost instruments in surgery technology have a very long life cycle. Howev...

Development of optimal packaging for medical...
Development of optimal packaging for medical products ENGINEERING In the contex...

Experience in apparatus engineering
The discipline of apparatus construction is very diverse and ranges from simple holding devices to c...
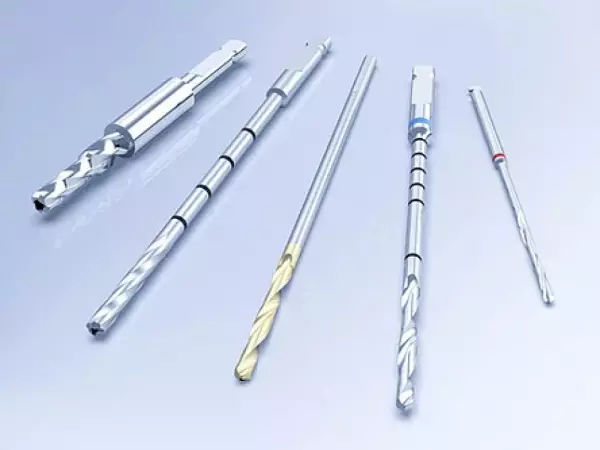
Orthopedic Products - Cutting Tools
The product range of cutting tools in the field of orthopedics includes: Bone drill Cu...

Modern diagnostic analysis methods require continuously smaller sample volumes. It brings advantages...

Milling technology / milled parts - contract...
Milling technology / milled parts - contract manufacturing for medical products Milling refers to...

The pure 2D CAD program AutoCAD is used by us for the creation of simple test drawings and the maint...

thoracic surgery - High-quality VATS instruments,...
High-quality VATS thoracic instruments, suction, retractors, electrodes - Made by Dufner Instrumente...

Development of medical devices, design of medical devices, development of products for medical technology companies, design of products for medical technology companies
The first phase of a medical product design and development project is the design phase. A mistake in design could affect the entire project and the product's life. A parallel activity that is designed to speed up the design phase can help the product develop at the quickest rate possible. After the design vision is agreed upon, the development process can move at full speed. Here are the four phases of medical device designing and development. You should follow each step in sequence.
Prototype testing is the final step in the design process. Testing of prototypes will identify any design issues and return the design back to the electrical, mechanical, and software team. Iterative modification is required in this process to ensure that the product is fit for purpose. This process must be repeated until the medical device meets all requirements, has been validated, and is ready for production. Iterative modifications are necessary for product validation.
The second step of medical product design and development is prototyping. This step involves the creation of a working prototype. A prototype is a scaled-down version of a device that has been tested. It is created in limited quantities. The process of producing a prototype typically involves one-off manufacturing processes, not cell manufacturing. However, with the advent of 3D printing technology, this process can be done much faster and cheaper.
During this stage, decisions need to be made regarding the manufacturing process. For example, the type of materials used must be chosen based on their durability and biocompatibility. The proper method of joining parts to each other is crucial. For example, the type of glue to use must be determined. The final stage of the design process is the assignation of each step of the manufacturing process. It is essential to identify the critical components of the product and determine how they interact with each other.
When designing a medical device, there are several steps that must be taken to produce the final product. The design process involves choosing the best materials for the components and choosing the right manufacturing process. The design process for the physical components of a medical device should be considered to be functional and safe. A successful project will meet the requirements of its users and its manufacturers. It will be a unique piece of equipment. The manufacturing steps are crucial to the success of a medical device.
During this stage, the product may be in its initial stage or a different stage. The design process should be as easy as possible for patients. If the product is difficult to understand, the user may be confused. It is important to understand that this is a necessary process. It is critical to have an excellent medical device. There are many other steps in the development and design of a medical device. The design phase starts with a conceptual prototype.
Become a digital exhibitor yourself in the online portal of the largest and best-known MedTech cluster region in Germany and inform the world of medical technology about your products and services as well as about news, events and career opportunities.
With an attractive online profile, we will help you to present yourself professionally on our portal as well as on Google and on social media.