
Metrological control of digital sphygmomanometers
Metrological control of digital sphygmomanometers Metrological control (MtK) on digital sphygmomanom...
Portal and digital medical technology fair of the largest MedTech cluster in Germany

Metrological control of digital sphygmomanometers
Metrological control of digital sphygmomanometers Metrological control (MtK) on digital sphygmomanom...

Environmentally friendly fluid management...
Biocide-free cooling lubricants with a long service life thanks to FluidWorker technology Water-m...

Automatic measurement on the CNC machine
Measuring on the CNC machine for more productivity hyperMILL® PROBING is the optimal and user...
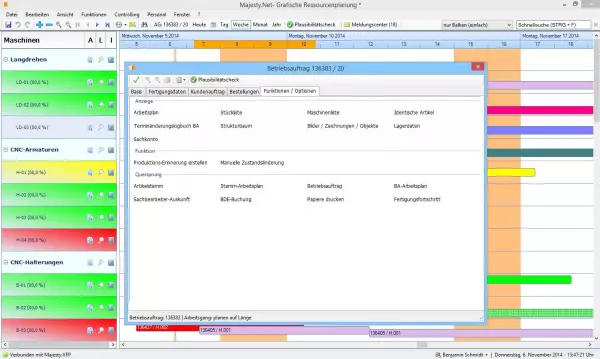
The perfect complement to the Majesty ERP software for medical technology and the machining industry...

Medical technology translations
Medical technology translations Our communication specialists have linguistic as well as technica...

Checking the health of medical devices
A staggering one in three medical devices have suffered recalls because of failures – with sof...

Cyber security for medical devices and medical...
Cyber Security The security of embedded devices is a pressing concern for society. Medical dev...
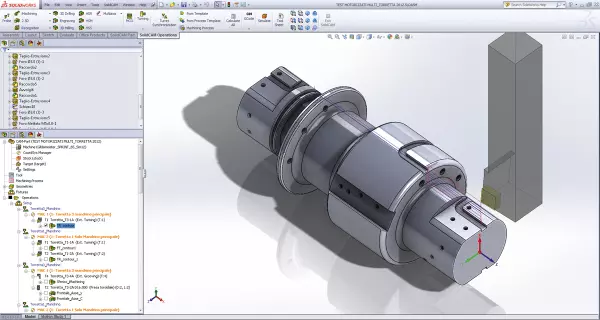
SolidCAM - The leading integrated CAM complete solution with the incredible iMachining technology...

Patent translation Your intellectual property is valuable. We help you to protect it internationa...

With the SOGA crimping press to the optimal result: The perfect hot crimping is a complex process...

EN ISO 13485 web documentation
Record changes on the website (can be used for EN ISO 13485) The DIN EN ISO 13485 is a quality ma...

CAD construction / development of medical...
CAD construction / development of medical products CAD has supplanted the drawing board and many...

Repair service for SpO2 sensors
We offer you various repair and service services for SpO2 sensors Repair service for your SpO2 se...

Additive manufacturing for medical technology
Additive manufacturing - only really perfect with CAD / CAM Additive manufacturing offers many ne...

GDSN Solutions for hospitals & GPOs
To share confidential content efficiently and consistently across Europe with hospitals, GPO's a...

Output increase in corona times
The plus10 AI technology increases the output of automated production systems on average by 10%....

Test lab services for the medical device...
The components and devices we manufacture must stand up to unrelenting physical, biological and chem...

Sell medical technology online with MAJESTY
Especially for medical technology customers, we have developed an interface to the award-winning e-c...

technology Majesty relies on the innovative and future-proof .net technology from Microsoft. A po...

Shannon - intelligent Operator Assistant
Our tool Shannon® continuously learns relationships between detected losses, most-likely root-ca...
Contract manufacturing of precise turned...
Contract manufacturing of precise turned and milled parts for medical technology perame...

Milling technology / milled parts - contract...
Milling technology / milled parts - contract manufacturing for medical products Milling refers to...

Quality-verification processes for the medical...
No industry faces a higher mandate for quality than medical device manufacturing. At ADMEDES, we und...

Higher quality finishing / finishing processes...
Higher quality finishing / finishing processes of surgical instruments / implants Refining or ref...

Metrological control of manual sphygmomanometers
Metrological control of manual sphygmomanometers Metrological control (MtK) on manual blood pressure...

The pure 2D CAD program AutoCAD is used by us for the creation of simple test drawings and the maint...
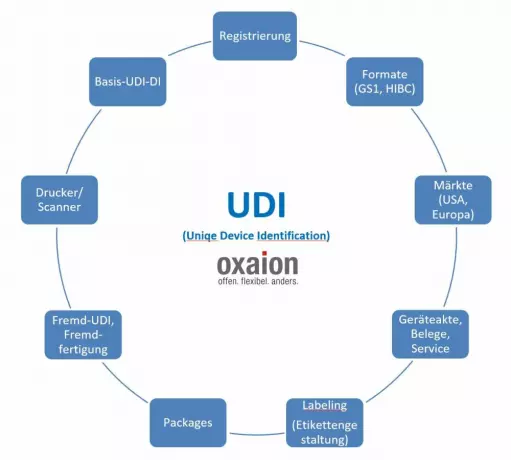
UDI as part of the business software in medical...
Not least because of the MDR, the UDI is developing into an essential component of overarching busin...

With the constant advancement in UDI databases worldwide, atrify is committed to supporting the impl...

Verification and validation of software in...
Our experience in a variety of safety-critical areas enables us to deliver an effective and efficien...

iMachining - The revolution in CNC machining Hold the knowledge and experience of hundreds of CAM...
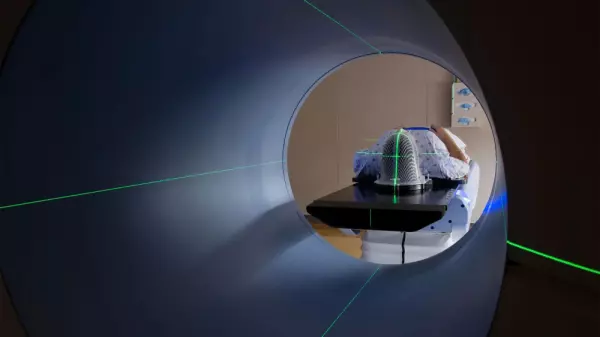
Development of Medical Devices - Systems...
Software Development We work in each phase of the medical devices’ development lifecycle an...
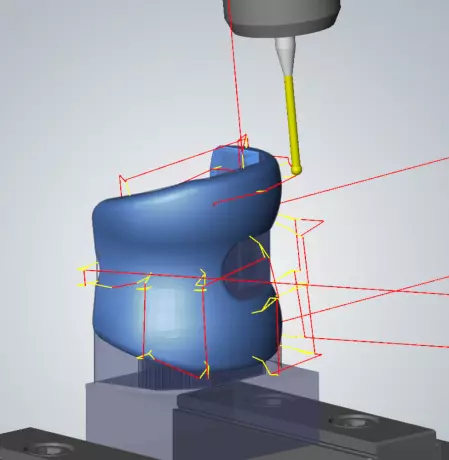
Intelligent component alignment for medical...
Intelligent component alignment at the push of a button Do you know the problem? You align the r...

Translation of technical documentation
Technical translation - Complex multilingual projects – This is exactly our thing Naturally...

Localisation of Medical Software
Software localization Software localization – developing and adapting software, online help...

The use of modern technologies opens up new possibilities in the treatment of complex defect situati...

oxaion easy medical technology - the business...
Smaller companies in medical technology in particular are feeling the pressure exerted by the new DI...
 Products and services from the categories Software, IT and cloud solutions / systems for medical technology in medical technology or other healthcare-related industries .
Products and services from the categories Software, IT and cloud solutions / systems for medical technology in medical technology or other healthcare-related industries .
Software for medical technology companies, software for the dental industry, software for laboratories, software for pharmacy, software for healthcare
Information technology (IT) is a rapidly growing industry in the U.S., which draws on a skilled and highly educated workforce of about two million people. These firms are renowned for producing reliable solutions, which benefit a wide range of industries. The strong protection of intellectual property (IP) laws in the U.S. has attracted international companies to the industry, which leads packaged markets and has a robust competitive landscape in almost all other segments.
Healthcare costs are rising all over the world, a situation that has spurred companies to develop health solutions. In 2016, a key trend will be increasing the efficiency of healthcare systems. Increasing the availability of remote healthcare is expected to contribute to cost reduction efforts. Big data collection and analysis will also contribute to tightening waste. In 2016, the healthcare software and IT infrastructure will support this trend. The use of IT for this industry will increase the number of doctors, hospitals, and insurers.
The application of software regulations reflects the fundamental tension between privacy and security, as well as health and safety. A high-risk software can lead to a harmful outcome for a patient. A low-risk software system may only help gather relevant information and inform treatment options. The application of these regulations depends on the context in which they are used. In critical contexts, timely action is vital to mitigate irreversible long-term consequences. The implementation of the rules should be consistent with the goals of the industry, including the protection of the health of people.
Medical devices are among the fastest growing industries. Most medical device companies are optimistic about the future of the industry in 2016. The market for medical electronics and healthcare IT was projected to exceed $100 billion by the end of 2015. The cost of healthcare is rising worldwide, and this will only increase as the population grows older. It is essential that medical IT software developers stay on top of the latest developments in the field. There are many challenges in the development of innovative health technologies.
The medical device industry is one of the fastest-growing industries in the world. Most medical device companies are optimistic about their future in 2016. With an ageing global population, healthcare costs are expected to rise, and medtech companies need new sources of funding. With Chinese investors, this is an opportunity that cannot be missed. The technology sector is constantly evolving. There are many different types of applications for healthcare. However, the main focus is on software and IT in healthcare.
The FDA has issued guidance on the classification of medical devices. This guidance provides criteria for software to be classified as a medical device. Examples of this classification are given in Box 2. These texts are also helpful for healthcare businesses. Most companies should be aware of the risks associated with these technologies. There are several factors that make software a good candidate for regulation. There are various benefits to using medical devices and IT. This is an essential part of any business.
Become a digital exhibitor yourself in the online portal of the largest and best-known MedTech cluster region in Germany and inform the world of medical technology about your products and services as well as about news, events and career opportunities.
With an attractive online profile, we will help you to present yourself professionally on our portal as well as on Google and on social media.