


Rudischhauser Surgical Instruments & Implants Manufacturing GmbH
Rudischhauser Surgical Instruments & Implants Manufacturing GmbH company portrait
YOUR PROFESSIONAL CONTRACT MANUFACTURER
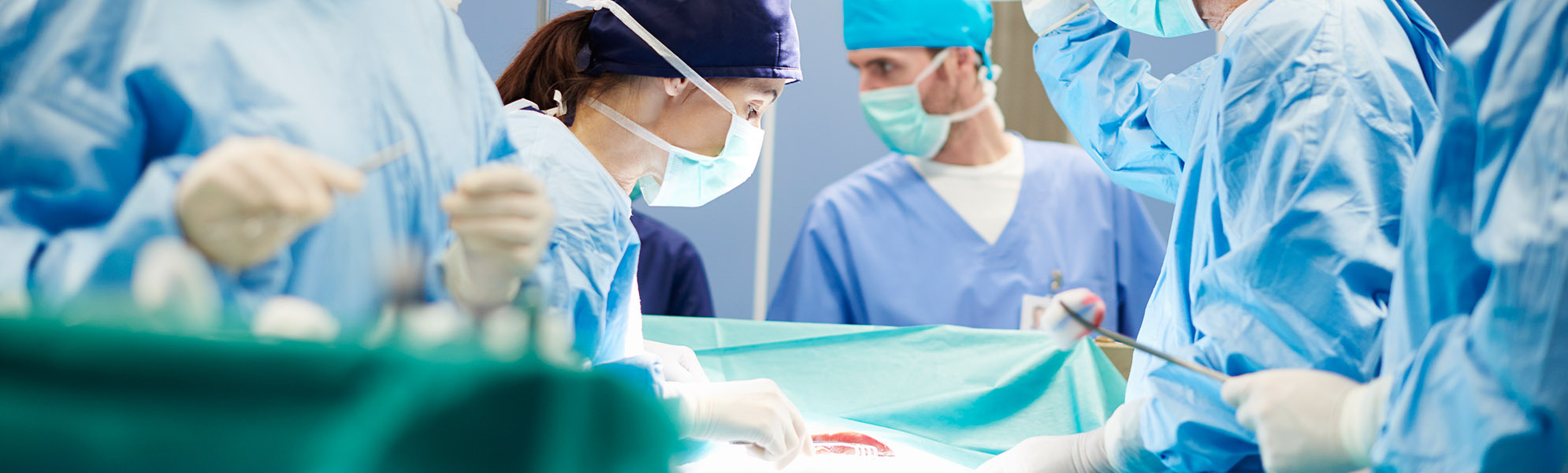
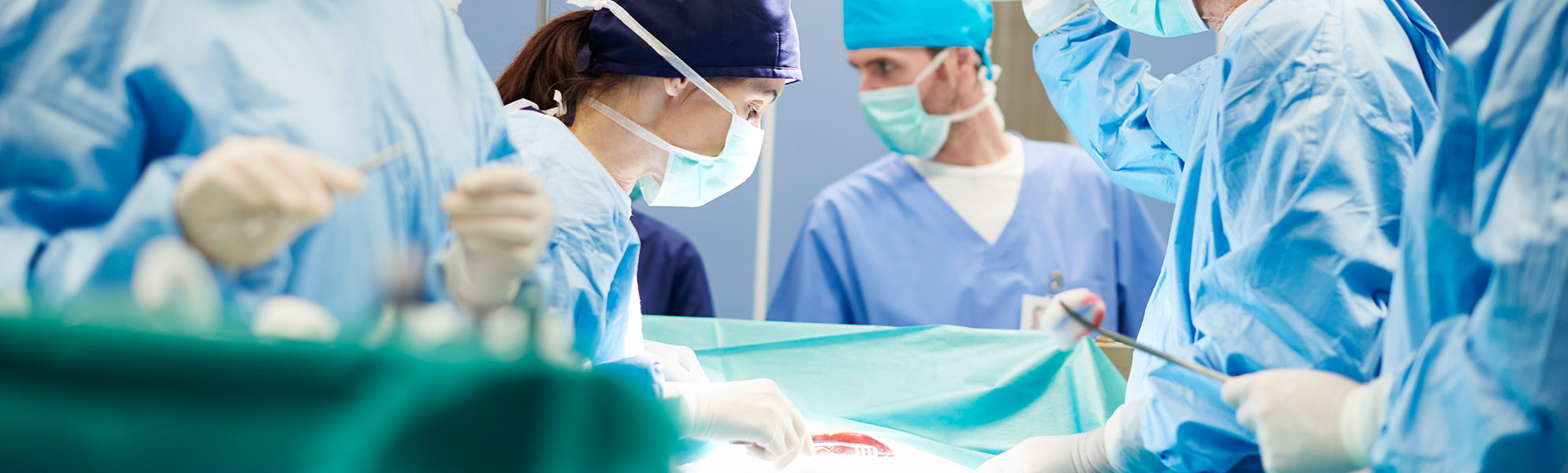
We are a contract manufacturer and development partner for high-quality instruments and implants in the fields of spinal surgery, orthopaedics and traumatology.
We also offer our customers complex, individual set configurations for spinal surgery complete with trays for instruments and implants.
MEDICAL PRODUCTS OF THE HIGHEST QUALITY
With our many years of experience and highly trained specialists and engineers, we have been developing and producing medical products of the highest quality using state-of-the-art development and manufacturing technologies since 1954.
THEREFORE RUDISCHHAUSER
-
State-of-the-art machinery
We work on 5-axis CNC machining centers and CNC sliding headstock lathes with up to 9 axes. -
Prototype production
We manufacture to the highest quality standards. -
Perfection down to the smallest detail
Our quality awareness extends into the µ range.
-
Expert support from A to Z
Our master surgical mechanics, cutting machine operators and toolmakers accompany each instrument from initial processing to final inspection.
-
From the model to the machine
We use the CAD data to program all milling and turning operations with SolidCAM - it couldn't be more efficient. -
3D coordinate measuring
Quality assurance with high-precision, validated 3D measuring machines and optical coordinate measuring machines.
QUALITY SINCE 1954
Rudischhauser Surgical Instruments Manufacturing GmbH, with its many years of experience and trained specialists and engineers, has been developing and producing medical products of the highest quality using the latest development and manufacturing technology since 1954.
With our own design and development department, we work closely with customers around the world to develop the optimum solution for each individual product (customized). With standardized and validated processes and procedures for your products, we meet the current legal requirements and thus ensure your competitiveness on the international market.
Special surface coatings, UDI-compliant laser marking and labeling according to customer specifications round off your product.
The company is certified according to DIN EN ISO 13485:2021 and registered with the US FDA (Food and Drug Administration) according to standard FDA 21 CFR, part 820 standard with registration number: 8010300.
AT A GLANCE
EMPLOYEES - 52 employees
EXPERIENCE - Over 70 years of experience in the manufacture of medical instruments
CUSTOMERS - 160 customers worldwide
INVESTMENTS - Over € 7.4 M Investments in the last 10 years
PRODUCTION AREA - Production area 1700 m² (18.298,6 ft²)
PRODUCTS - Own product line consisting of instruments and sets
TECHNICAL R&D SUPPORT - CAD/CAM department (SOLIDWORKS & SolidCAM)
CERTIFIED - DIN EN 13485:2021 certified for the production of surgical instruments and implants FDA registered
INSTRUMENTS & IMPLANTS - 45% Spine, 25% Orthopaedics, 25% Traumatology, 5% Other
FACTS AND FIGURES
- CNC sliding headstock automatic lathes: 9 machines
- CNC machining centers: 6 machines
- Quality control: 4 employees
- Technical support: 4 employees
- Glass bead & granulate blasting: 4 blasting machines
- Laser marking | UDI: 2 Laser Marking Systems
- Laser welding: 2 laser welding systems
- Polishing: Manual | Electrolytic
- Coating | surface treatment: DLC | AlTiN | titanium anodizing
- Final cleaning | Labeling | Packaging
DEVELOPMENT
INDIVIDUAL AND CUSTOMIZED
In close cooperation with the customer, we develop, optimize and produce standard and special instruments, set configurations and complex assemblies including assembly, laser engraving and labeling.
YOUR ASSISTANCE IS NEEDED!
Do you have a product which could be optimized? An idea or even vague concepts or suggestions for a new product? Simply give us a call or write to us. We love accepting any challenge.
TYPICAL PROCEDURE
- Problem definition | Target
- Brainstorming
- Design | Drawing
- Prototype construction
- Serial production
PRODUCT OVERVIEW
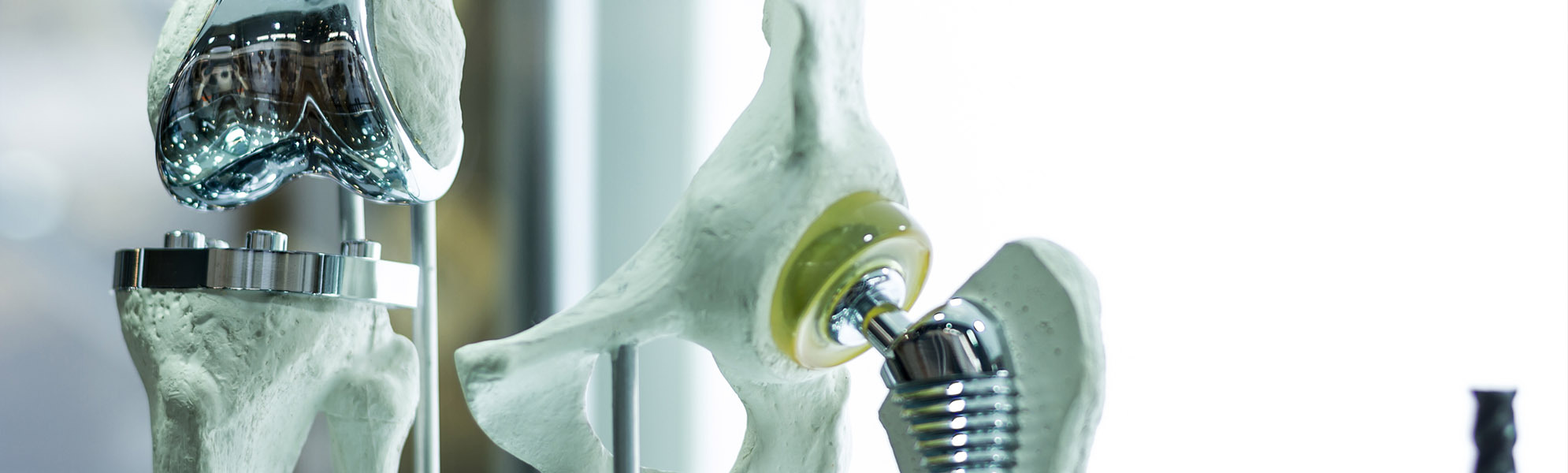
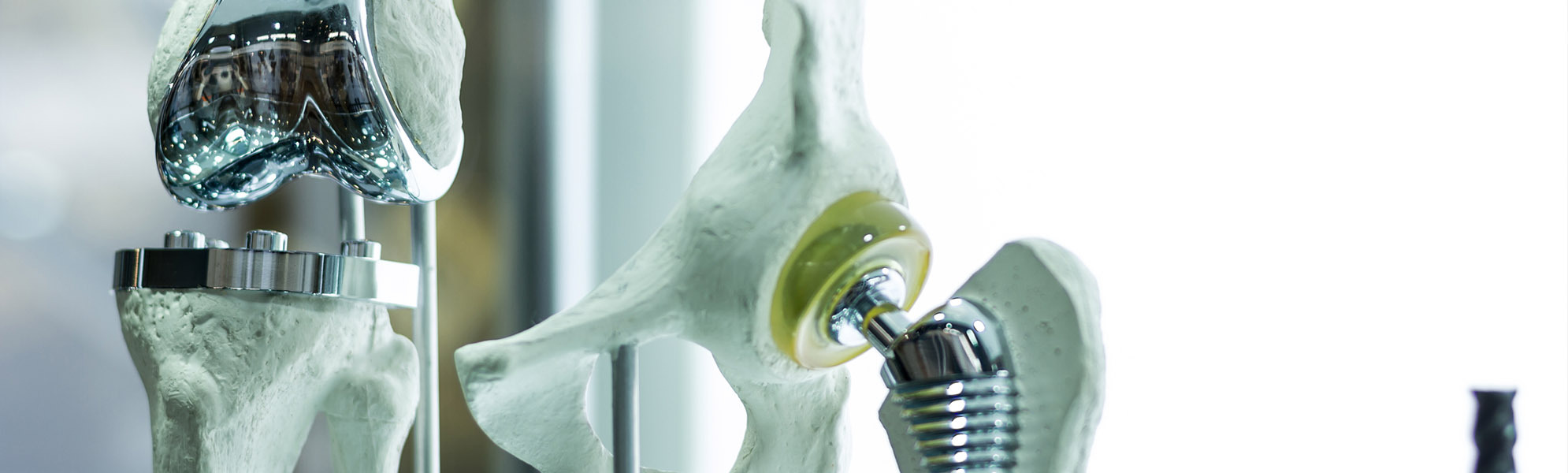
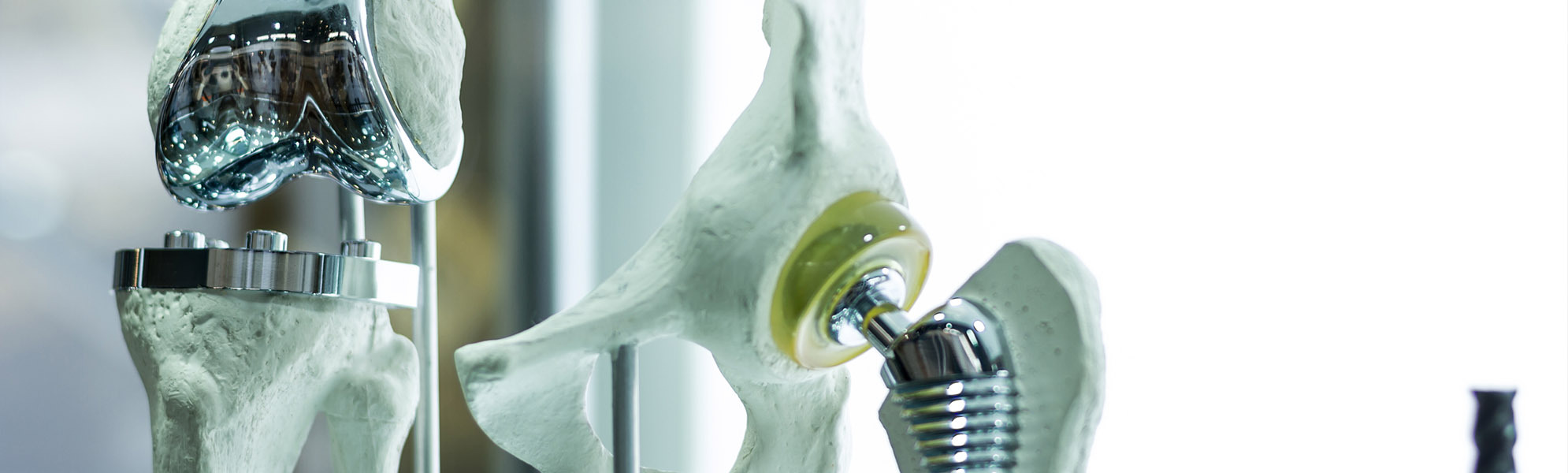
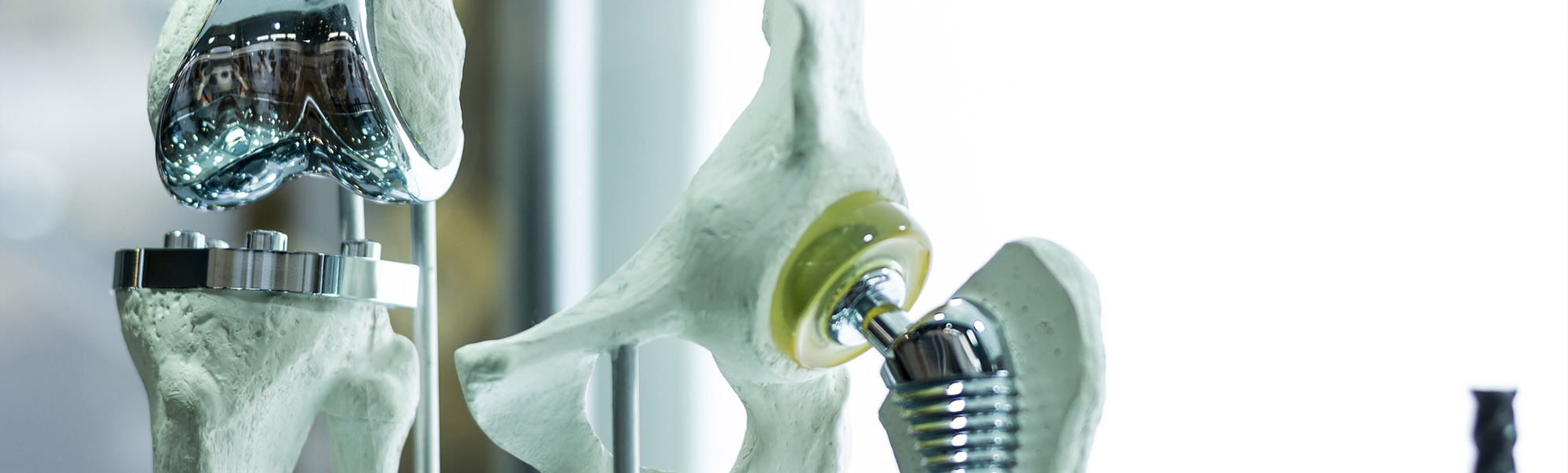
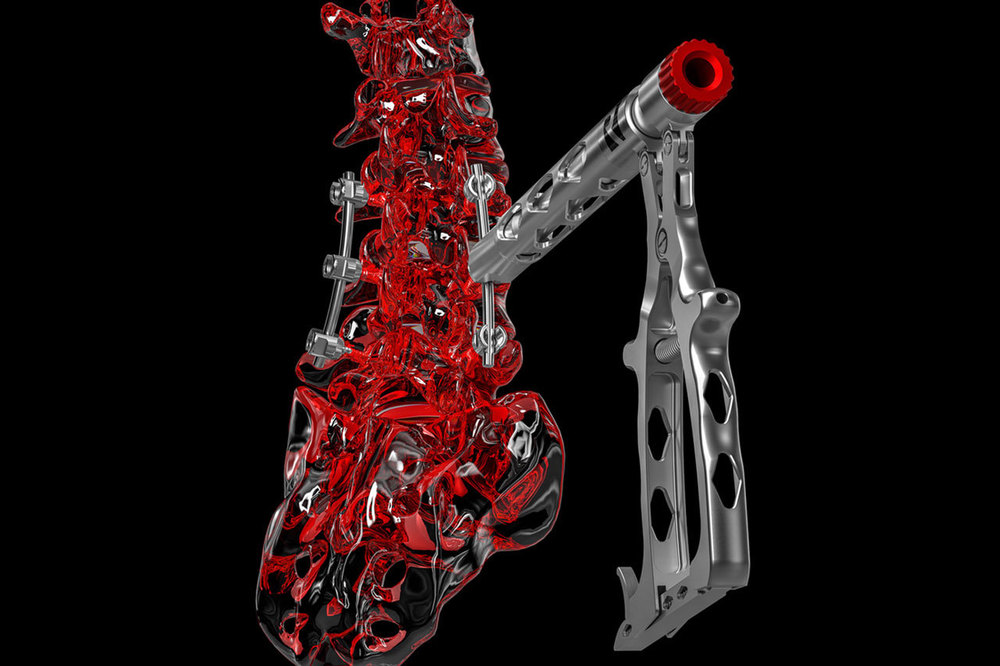
SPINE
Development, optimizing and production of high quality products such as implants ranging from cervical to lumbar, made from widely diverse materials, and a broad variety of different instruments which, regardless of the indication involved, are available in options ranging from an individual instrument or complete set solution (with or without silicone handles) to a product provided in sterile packaging, including the documentation required by regulations.
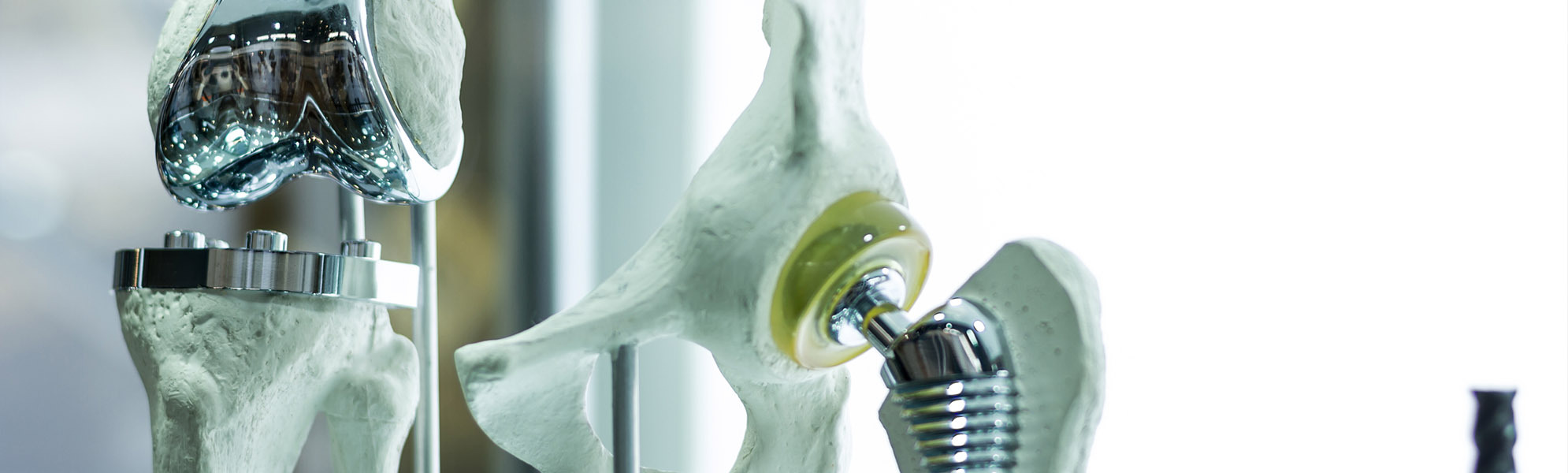
ORTHOPAEDICS
Innovative solutions for every joint region, such as the hip, knee and foot. Here we offer instruments and complete sets. In close cooperation with our customers, the desired requirements are collected, recorded and then implemented by our qualified specialists to create a high-quality medical product.
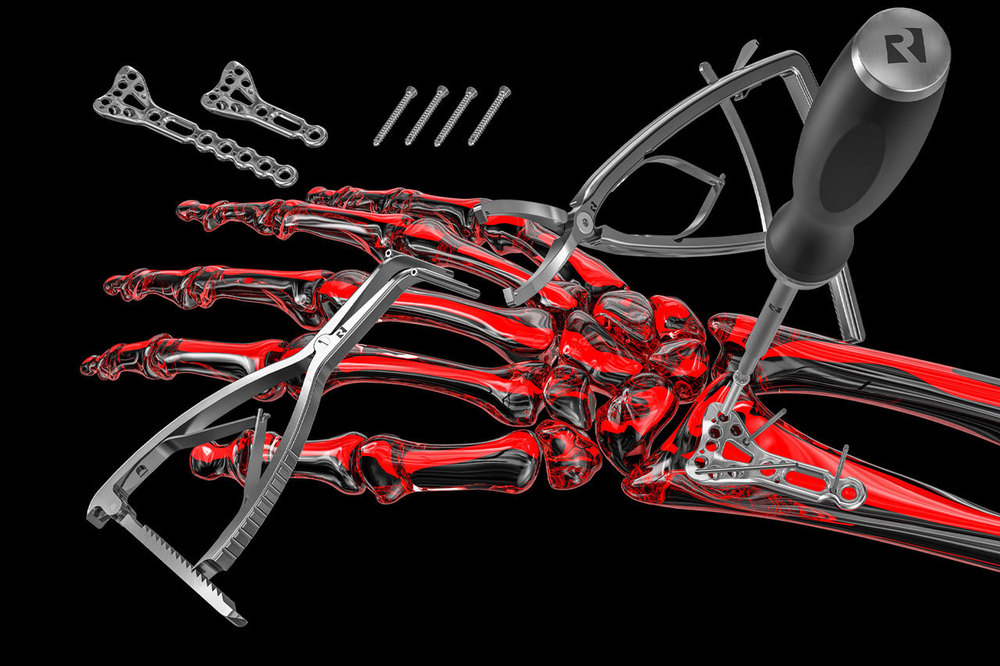
TRAUMATOLOGY
In the field of traumatology, we develop and produce an extensive portfolio for internal and external fixation. Whether small or large fragments, we offer expertise from the initial idea to the production of instruments and implants as well as complete set configurations.
Rudischhauser Surgical Instruments & Implants Manufacturing GmbH > Zertifikate
Zertifiziert nach DIN EN 13485:2021 und registriert bei der US-Amerikanischen FDA (Food and Drug Administration) nach Standard FDA 21 CFR, Teil 820 Standard mit Registrier-Nr.: 8010300.> Diese Kategorien könnten Sie interessieren

Rudischhauser Surgical Instruments & Implants Manufacturing GmbH
Gänsäcker 2878532 Tuttlingen
Deutschland
Fon: +49 7462 945629-0